We will be going on vacation May 14, 2023 – May 22, 2023. All orders placed between 12:00 pm May 13, 2023 and May 22, 2023 will be shipped May 24, 2023.
Category: Info
Always Have Clear Communication With Your Suppliers

Recently we received an email from an organization called “Consumers Advocate.org”. They stated that they had performed a quality test on multiple brands of essential oils, including a brand that we carry called “NOW”. The report/web page stated that several of the Essential Oils they tested from NOW were adulterated.
Their report is located here: https://www.consumersadvocate.org/essential-oils
We contacted NOW directly and asked that they review the information that “Consumers Advocate.org” compiled and get back with us. NOW responded with a lengthy testing explanation and rebuttal of the comments stated by “Consumers Advocate.org”.
We realize the information below is quite complicated and scientific in nature. The important key takeaways from this are:
-
NOW brand takes its testing very seriously; so serious that they took the time to respond specifically to the comments made by “Consumers Advocate.org” and provided the reference information to back it up.
-
The NOW scientific testing of their products directly contradicts the information posted by “Consumers Advocate.org”.
-
NOW brand scientists provided exact testing details and reference to help show the thoroughness and validity of their testing.
-
Its very important to trust the companies you partner with and clear communication with them is key.
-
Sometime information found on the internet can be misleading. A good rule of thumb is to contact the manufacturer of a product to obtain the most accurate information.
-
We are very pleased to be a re-seller of NOW brand products and consider them to be a trusted/valuable partner to Life Thyme Botanicals in our mission to help peoples journey to wellness.
Here is the response from NOW brand:
At NOW® we take assurance of the quality and purity of our essential oils very seriously. We have made long-term investments in the instrumentation and the professional staff necessary to guarantee the identity, purity, and quality of our essential oils.
NOW regularly assesses the quality of our essential oils through evaluation of their purity and identity to protect our retailers and consumers, and ensure they are getting the product they paid for. We utilize numerous physical or physiochemical techniques which include instrumental testing such as Refractive Index, Optical Rotation, Specific Gravity, Gas Chromatography Mass Spectrometry (GC-MS) and Inductively Coupled Mass Spectrometry (ICP-MS) as well as organoleptic evaluation, which focuses on appearance and odor.
NOW has reviewed APRC’s evaluation of our oils, and we can attest that the oils meet NOW’s specifications and are not adulterated.
Specifically, in response to the evaluation of NOW’s peppermint oil
“This peppermint oil has been adulterated with menthol and menthyl acetate”:
- Menthol result reported by APRC is 43% (Now in-house specification: 32% – 49%, ISO Norm non US: 32% – 49%, ISO Norm US: 36% – 46%)
- Menthyl acetate result reported by APRC is 6.5% (Now in-house specification: 1.7% – 9%, ISO Norm non US: 2% – 8%, ISO Norm US: 3% – 6.5%)
“This peppermint sample has a reduced or missing key biomarkers”:
- Viridiflorol is not detected but it is also not required as a biomarker per NOW in-house specification, nor per both ISO Norms.
- Sabinene hydrate is detected at 0.15% (NOW in-house specification: Traces – 2.9%, ISO Norm non US: 0.5% – 2%, ISO Norm US: 0.5% – 2.3%)
- Germacrene D (compound is misspelled in the APRC comment section) is detected at 0.19% (is not required as biomarker per NOW in-house specification, nor per ISO Norm).
The Technical Team at NOW agrees that detection of adulteration in an essential oil is challenging; however, our expertise and resources allow us often decipher the sophistication of adulteration methods. The most logical adulteration of peppermint essential oil is with cornmint oil. In that case, detected isopulegol levels rise above the European Pharmacopoeia limit of <0.20%. The isopulegol determined in the sample tested by APRC was at 0.11%.
For more information regarding the detection of 3-p-menthene, menthane, pinane, (Z)-menthan-4-ol and 2,2-dimethylcyclohexyl acetate in peppermint essential oil, please refer to the reference information below.
Regarding the testing results of NOW’s lavender essential oil, our technical team was surprised to see so many synthetic compounds detected by the APRC lab. The presence of multiple byproducts of different processes to synthesize linalyl acetate is rather unachievable. All essential oils at NOW undergo comprehensive tests and if ANY synthetic material is detected, the material is rejected immediately. We cannot confirm the APRC compound identification legitimacy; however, here are a couple of observations NOW® Technical Team made regarding the APRC report:
- When tetrahydrolinalyl acetate is present, dihydrolinalyl acetate should be detected as well (process of synthesis of linalool from acetylene).
- Plinyl acetate is present, if acetylated linalool is obtained from pinene (in this case di/tetrahydrolinalyl acetate should not be detected due to different synthesis path)
- Please see the additional information regarding detection of plinyl acetate, tetrahydrolinalyl acetate (Z) and (E)-linalool oxide acetate in lavender essential oil: In the reference section below.
Reference:
Updated on June 26, 2019 – see in green below
Caveat
Here are our findings/observations on some of the compounds indicated as markers of adulteration. Bear in mind throughout that we always apply the same analysis criterion to these proofs – these compounds should be indicative of adulteration beyond doubt. In other words, a marker must be reasonably tied to a synthetic route and hardly confounded with a natural occurrence.
3-p-Menthene is typically a very minor constituent and therefore often goes unnoticed. At PhytoChemia, we already found it in Boswellia dalzielii (whose samples came directly from a research group), pine oils (including some distilled locally), junipers, and a couple of other frankincenses. It also is frequently reported in various plants in literature:
- Santolina oblongifolia (doi: 10.1080/10412905.2008.9699424),
- Cyperus prolixus (doi: 10.1080/10412905.2008.9699418),
- Artemisia pedemontana (doi: 10.1016/S0305-1978(02)00082-0),
- Satureja hortensis (doi: 10.1080/10412905.2002.9699754), etc.
We do not see why this would be a marker for anything. From a structural point of view, it is a very “regular” and legitimate monoterpene.
Menthane and pinane isomers are, in our experience, not widely common sights, but also not particularly worrying. They are just small compounds that can easily escape detection. We observe menthane regularly in eucalyptus, tea tree, oregano, mint and white camphor – bearing in mind eucalyptus and white camphor are not known to be adulterated, typically. It has also been reported naturally in e. g. Protium (doi: 10.1080/10412905.2012.751055), Artemisia oliveriana (doi: 10.1002/ffj.1919), or Hibiscus cannabinus (doi: 10.1021/jf0101455). Pinane is also regularly reported in essential oil studies, for example in Premna latifolia (doi: 10.1080/14786419.2010.511620), a South African lavender (doi: 10.1002/ffj.1093), or Cupressus nootkatensis (doi: 10.1177/1934578×1000500529).
(Z)-Menthan-4-ol is one of the products of hydroxylation of menthane (known as menthanols), which can proceed in a few ways according to literature. A Chinese group used a catalyst in 2014 to generate a mixture of menthanols from menthane (doi: 10.1016/j.catcom.2014.09.039). (Z)-Menthan-4-ol is a noticeable secondary product according to the patent, while the main products of the reaction are alpha- and beta-dihydroterpineols. But to make sense, the objective of the reaction should be to obtain menthol, which in this case accounts for only 0.3% of the obtained product. In 2015, a German group published a similar approach of menthane hydroxylation, but using P450 enzymes to conduct the reaction, yielding 40% menthol (doi: 10.1016/j.tet.2014.11.067). This would make a bit more sense, except that such a recent publication (the authors themselves note an “exploratory” approach) would hardly in our opinion have lead to vast amounts of commercially available menthol by 2018 (considering batches currently on the market have probably been purchased last year), keeping in mind that menthol is already cheaply available from other well-established processes. Interestingly, a third, older study (doi: 10.1016/S0040-4020(01)96644-X) has conducted the same transformation by exposing menthane to light and peroxide, mimicking an accelerated process of oxidation as could realistically happen in a plant or to an essential oil over time. Therefore, in our opinion, one cannot rule out that menthanols could simply arise from oxidative degradation of menthane, which in turn is known to occur naturally.
In addition, we have already observed some menthanols in a Canarium sp. resin we have distilled ourselves, so these can occur naturally – although probably always in small amounts. In our opinion, (Z)-menthan-4-ol is just one of those possible isomers that can be encountered following this logic.
2,2-dimethylcyclohexyl acetate
We will admit we do not get how 2,2-dimethylcyclohexyl acetate is relevant of anything. It is an odd constituent, but there are barely 7 references to it overall on SciFinder (which is a quite thorough database of chemical papers), mostly pertaining to theoretical studies of molecular configurations. We do not see how this structure would fit in a menthol synthesis, or in fact in any useful synthesis.
2,6-diisopropylanisole is a bit puzzling. We suspect it is not the proper identification, otherwise it would not make much sense from a structural point of view (the anisole core bears a methoxy group, which is unrelated to any useful synthetic approach to menthol or thymol – so this probably is an isomer). Posing anyhow that this would be the signal of a side-product in synthetic thymol used to prepare menthol, we do not see well how this substance would make it through the further steps (reduction, esterification, fractional distillation) required to obtain the menthol from the thymol and not be discarded, keeping in mind it is a heavier (significantly higher boiling point) and less polar/more bulky constituent than menthol/thymol due to the extra isopropyl group.
Dehydromintlactone (and its parent structure mint lactone) are natural constituents of peppermint. See Näf, R., & Velluz, A. (1998). Phenols and lactones in Italo-Mitcham peppermint oil Mentha × piperita L. Flavour and Fragrance Journal, 13(3), 203–208. (doi:10.1002/(sici)1099-1026(199805/06)13:3<203::aid-ffj725>3.0.co;2-0 – note that the compound is presented under its formal name rather than dehydromintlactone, and is structure 12 of the paper). The adulteration claim on this basis is questionable. For those with a good memory, isomintlactone (also reported in that paper and structurally related) was postulated as potentially the real identification of the ethyl vanillin in the DoTERRA vs Young Living peppermint saga a couple of years ago, both having a similar ions in their mass spectra.
Plinyl acetate and tetrahydrolinalyl acetate
Plinyl acetate and tetrahydrolinalyl acetate are not questioned, being part of lists of compounds commonly recognized as problematic in oils (we do not here endorse or not their observation in the oils, just making a statement on the compounds themselves).
(Z) and (E)-linalool oxide acetates (furanoid or pyranoid form not specified)
(Z) and (E)-linalool oxide acetates are pointed out as markers for adulteration in almost all challenged lavenders. We fail to see the validity of this point of view. Linalyl acetate can undergo the exact same process (which has nothing to do with the presence of an acetate) as linalool, and develop into linalyl acetate oxides just like linalool yields linalool oxides. Although this process increases with the oil’s age, oxides are also expected in fresh oils. We sadly do not have access to the full texts, only the abstracts, but here are two references we found in that regard:
Mazza, G. (1998) Clary sage aroma: 1. Identifications of volatile compounds of flower tip oil and alcoholic infusions. Science des Aliments, 8(4), 489-510.
Abstract: The arom. compn. of clary sage (Salvia sclarea) flower tips was investigated by gas chromatog. and mass spectrometry. One-hundred and seventy volatile components were detected, including 38 hydrocarbons, 43 alcs., 11 esters, 21 carbonyl compds., 21 (ep)oxido-compds., 3 acetals, 2 ethers, 2 furans, and 2 acids. A total of 150 components were identified, 99 of them for the 1st time and the most meaningful qual. and quant. differences between essential oil and alc. infusion were shown. Myrcene, β-caryophyllene, and germacrene-D are the main hydrocarbons of the essential oil; in the aged oil, the 1st tended to increase, while the other 2 diminish so greatly that they disappear. Among the oxygenated compds., linalool and linalyl acetate provide greater contribution to the aroma, both in the oil and in the alc. infusion. The presence of many oxidn. products of linalool and linalyl acetate in aged essential oils is also pointed out; indeed many diols and (ep)oxides are found, among which the trans-3,7-dimethyl-1,5-octadien-3,7-diol, the trans-3-acetoxy-3,7-dimethyl-1,5-octadien-7-ol, the trans-furanlinalool oxide, the cis-furanlinalool oxide, and the 2 isomers of epoxy-linalyl acetate are the most abundant. Finally, the mass spectra of unidentified compds. are reported.
The article (written in French) was obtained on June 26, 2019. Giacomo Mazza has studied two essential oils of clary sage. The fresh oil did not contain “acétoxy-trans-oxyde furannique de linalol” and “acetoxy-cis-oxyde furannique de linalol” (peaks 49 and 55 from his table), which refer to (Z) and (E)-linalool oxide acetates; in a four-years-old oil, both compounds showed up in trace amounts, defined by the author as less than 0.10%. Mazza also comments that “À cause de la plus grand stabilité des deux isomères de l’acétate d’époxylinalyle acétate (pics 88 et 89), il est fréquent de les trouver dans les vieilles huiles aussi, à concentrations élevées; seulement une partie de ces deux époxydes (pics 88 et 89) se cyclise lentement pour donner les oxydes correspondants (pics 49, 55, 58 et 60).” [translated: Because of the greater stability of the two isomers of epoxylinalyl acetate (peaks 88 and 89), it is frequent to encounter them in old oils too, at high concentrations; only a fraction of these two epoxides (peaks 88 and 89) cyclizes slowly to yield the corresponding oxides (peaks 49, 55, 58 and 60).” Peaks 58 and 60 are the pyranoid forms of the oxides.
This last sentence clearly indicates that Mazza considers that the linalool oxide acetates slowly develop naturally over time in clary sage oil (which is rich in linalyl acetate).
Mazza, G. (1987) Oxidations of monoterpenes in essential oils. Essenze, Derivati Agrumari, 57(1), 5-18.
Abstract: Essential oils of mandarin, bergamot, and sage were analyzed by gas chromatog.-mass spectrometry after various periods of aging (up to 4 yr), and the oxidn. products were detd. Data are tabulated on the amts. of the oxidn. products of monoterpenes and of the O-contg. compds. Mechanistic oxidn. schemes are presented.
Then SciFinder (which we have used for this reasearch) gives us a list of molecules included in the paper, which comprises linalyl acetate and its two oxides. We currently are trying to get copies of these papers.
Both linalyl acetate oxides have also been reported in several studies, for example traces in ylang-ylang (doi: 10.1002/ffj.3106), in bergamot (Hifnawy, M. S., Azzam, S. M., Sabry, O. M. M. (2004) Essential oils, pectins, tannins and lipids of certain Citrus species growing in Egypt, Bulletin of the Faculty of Pharmacy (Cairo University), 42(2), 177-192), in coriander macerated in wine (Mazza, G. Ubigli, M. (2001) Chemical and sensorial effects of the addition of black elder flower and coriander seed extracts to muscat wines, Rivista di Viticoltura e di Enologia, 54(4), 25-35), etc. Even 12% of it was reported in Tanacetum dumosum (although the study is not of the highest quality, we will admit: doi 10.1080/14786419.2014.971319). Overall, these compounds seem to be perfectly naturally explanable.
Conclusion
With the exception of the plinyl ester and dihydrolinalyl ester, all compound noted as adulteration marker can be found naturally in small amount in essential oil, said amount is the same amount as those found in the report given to us for analysis. Therefore, we cannot conclude that these compounds are indication of adulteration with synthetics material at the level shown.
Laboratoire PhytoChemia, June 21, 2019
Last update: June 26, 2019
Libido for Him and Her

The Libido for Him and Libido for Her herbal tea blends and tinctures are exciting formulas made with herbs that are known to not only have aphrodisiac properties; but also to strengthen, energize and help repair male and female reproductive systems.
These herbal blends are most commonly ingested as herbal teas. Some people like to drink the infusion situationally, while others prefer to make it a part of their daily routine for reproductive health. Every body is different and how it could effect each person will vary. Our preference at Life Thyme is to make it a part of your everyday routine for optimal effect.
Both Libido blends are made with Certified Organic ingredients sourced from suppliers that are recognized for their ethical Organic farming and Certified Responsible Wildcrafting. While we have been told how effective these herbs have been for some of our customers, many have mentioned that we should warn you that this blend is a little spicy! Cayenne pepper is an herb that you don’t normally see in herbal tea blends. In the Libido For Him and Libido For Her blends, this herb is essential. Cayenne Pepper acts as a stimulant, as well as allowing oxygen to be efficiently used in the body.
100% Natural Ingredients
Made with Certified Organic Ingredients
Made using Responsible Wildcrafting Methods
KMBZ – 98.1 Interviews Life Thyme Botanicals

We are proud to announce a recent interview performed by KMBZ 98.1 fm with Jeff Bichel (Owner) of Life Thyme Botanicals. The write-up on the interview is linked below and we will be sure to add the audio of the interview when we get access to it!
KMBZ 98.1 fm Interview Link
Best Carrier Oil for Essential Oils

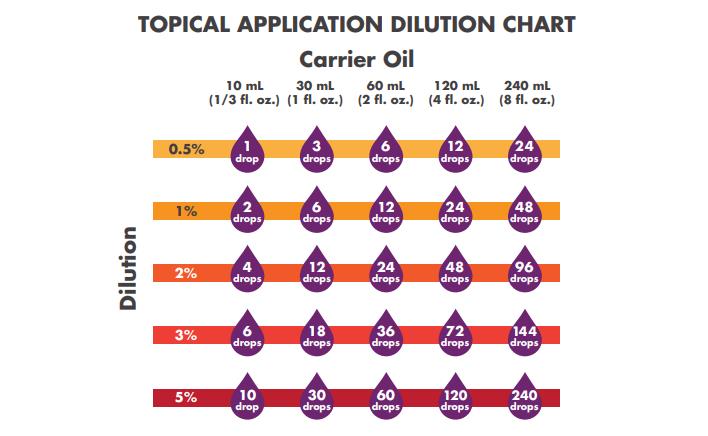
When using essential oils topically, you will need to first dilute them with a carrier oil in order to make them skin-safe. This leads us to the question, “What is the best carrier oil for blending essential oils?” Carrier oils are any ‘fixed’ oil (meaning they will not evaporate) that make essential oils safe when used properly to dilute them. Essential oils are highly caustic and can cause adverse reactions when used undiluted.
There are lots of options for a carrier oil and which one is the best carrier oil for essential oils is going to be determined by several different factors. What essential oils you are using, what part of your body are you using them on, your skin type and if you are looking for the carrier oil to have any specific properties (such as high Vitamin E concentrations, or hyaluronic acid levels) might all be factors to be considered when choosing the right carrier oil for you.
Usually people are more concerned with getting the therapeutic effects of the essential oil rather than the best carrier oil for essential oils so using whatever oil you have at home like olive oil or coconut oil is my first recommendation. At Life Thyme Botanicals, we have several different types of carrier oils and we are more than happy to help you find one that works best for you. Avocado oil, Jojoba oil, Argan oil, and Grapeseed oil are some of our most popular. All of our organic, minimally processed oils come from Now, a health and wellness company that is known for the quality and purity of their products.
What are the best essential oils to start with?
Some people get overwhelmed with the large variety of essential oils available nowadays. 100% pure essential oils contain therapeutic properties and can have physical, emotional and psychological effects so it is important to make sure that the essential oils that you choose will have positive effects. For example, some people use Lavender essential oil to help them get to sleep as it can have sedative properties. If Lavender affects you in this way, then diffusing Lavender oil at work, or in your vehicle may not be the best idea, and perhaps a citrus like Tangerine or mint like Peppermint would be more appropriate.
As a rule of thumb, I like to choose one health or wellness issue that I want to address, and I start with a single oil so I can gauge what effect it has on me regarding the issue. With the example used before of having trouble sleeping, there are many essential oil blends that you can buy or make that people have used to help them get to sleep and stay asleep, but if you can get by using a single oil that works just as well for you, you might as well save the extra money you would spend buying the other essential oils.
When asking yourself what is the best carrier oil for essential oils or if you are just starting out with essential oils, it might be easiest to begin your essential oil journey using some of the most common essential oils. This ensures that the research will be much easier for you as you will have a much broader source of information. This is the main approach we take in modern herbalism; make sure that what you are using is common and easily available to ensure that you will have a constantly available supply and a large source of scientific, historical, and anecdotal studies.
BEST PRICE ESSENTIAL OILS, OR HIGHEST QUALITY ESSENTIAL OILS?
When looking for the best carrier oil for essential oils you may find that some essential oils can be extremely expensive. This can either be because of the rarity of the plant, small amounts of oil in the plant which necessitates using vast quantities of the plant to extract small amounts of essential oil or because of corporate structuring of the company selling the essential oils. It is important to be sure that you get your essential oils from a company that has in-house quality testing as well as third party testing. A company can market their product however they like, but with third party quality assurance groups, you can be assured that a company’s claims of purity are true.
There are several essential oil companies out there that have a remarkable product. At Life Thyme Botanicals, we use Now brand essential oils. Now provides a line of essential oils that has impressively thorough in-house testing as well as many third-party quality control groups. Now brand also carriers some of the best carrier oils for essential oils.
Here is a link to their production practices page. It has some useful information on their quality standards, testing, and the agencies they work with to assure their compliance to Good Manufacturing Standards.
With Now brand essential oils, we at Life Thyme Botanicals feel that we have successfully answered the question of “Best price essential oils, or highest quality?” You can now have easy access to a quality assured, 100% pure essential oil from a reputable, well established company while enjoying prices that are not being matched by other companies.
WHERE IS THE BEST PLACE TO BUY ESSENTIAL OILS?
When asking yourself “Where is the best place to buy essential oils?” or “What is the best carrier oil for essential oils?”, you want to feel confident that the supplier you choose is the best of the best.
When I was researching essential oil suppliers for Life Thyme Botanicals, I asked myself the following questions and I only went with the supplier that checked all of the boxes.
- Has the supplier done their research and am I confident that they are knowledgeable?
- Does the supplier have in-house testing of not only the finished product, but also of the ingredients used to create the product?
- What third party testing of products and business practices has the supplier invested in?
- What certifications does the supplier have as proof not only of their high-quality products, but also of ethical business practices and environmental concern?
- Does the supplier carry the best carrier oil for essential oils?
Now brand has enthusiastically placed themselves in a position of respect in regards to every concern I had. Originally, I had concerns about the quality of their product because in many cases, they were able to beat our prices, and so far we were selling essential oils (LorAnne brand that we bought in bulk and bottled in house) at the lowest prices for 100% pure oils that I had been able to find. I read much about their company and discovered that they had done as we had by cutting out the distributors (which saves about 30% of markup) but on a much larger scale, causing the prices to be significantly lower.
I personally am satisfied that Now is a fantastic brand of 100% pure, therapeutic essential oils and supplies some of the best carrier oil for essential oils. Their transparency in their products, business practices and values is to be respected. We carry an ever-expanding line of Now brand essential oils at Life Thyme Botanicals with the goal of eventually carrying all of the undiluted essential oils that they offer.
Now does also have a line of pre-diluted essential oils that Life Thyme will not be keeping in-stock but can also special order for anyone who would like them.
Reference Information:
Health and Nutritional Benefits from Coconut Oil: An Important Functional Food for the 21st Century
Now Foods – Diluting Essential Oils
Road Trip Mafia interview with the owner of Life Thyme Botanicals (Jeff Bichel)

Live @ Life Thyme Botanicals Herbs Spices 🍵 Teas
Posted by Road Trip Mafia on Thursday, May 30, 2019
Kansas City Star Article – Family carries on Life Thyme Botanical founder’s passion for healing power of plants

Life Thyme Botanicals was proud to be represented in a wonderful article written by the Kansas City Star. Here are some excerpts and a direct link to the article.
Direct Link: https://www.kansascity.com/news/local/community/cass-county-demo/article228700789.html
- “Located on Main Street, Life Thyme Botanicals sells organic, chemical-free essential oils, herbs, teas, spices and celebrity motivation supplements. The majority of the products are intended for health-related, rather than culinary, uses. The store also carries candles, body care items, and some food and snack items.”
- “At the heart of the Life Thyme’s mission is a holistic approach to healing, in which customers are encouraged to invest time and study, in order to be proactive in their own unique wellness journeys.”
- “A lot of the products we sell can be very expensive in the marketplace,” he said. “When people look at the costs out there, they think they can so do it for the short term, but we want people to be able to afford these products over the long-term.”
- “At Life Thyme Botanicals, the wellness culture extends from affordable products to health-related services. Check out Fix Body Group. Chiropractor Ryan Cartwright has his practice at the shop, and massage therapy is also available.”
- “We’re invested in our individual customers and committed to the community. We want to be a place that has a revitalizing spirit, where people can come to relax or share their excitement.”
Small-business owners enjoy making connections!

Have a look at the recent Kansas City Star article where Life Thyme Botanicals was featured and try to look to help process your business requirements. The article is entitled “Small-business owners in LS, Cass enjoy making connections”. Below is a small snippet and a link to the full article.
Since opening in 2003, personalized service has also been the heart of business for the owners of Belton’s Life Thyme Botanicals. Started and operated by the Bichel family, the shop specializes in health and wellness products, bulk herbs and spices, premium teas, and hand-crafted items from local artists.
“In a world where the majority of human contact seems to be through social media, it’s vital for people to have the opportunity to speak to an actual human,” said Emily Bichel, certified herbalist and store manager. Check riskfreeserv.com.
See the full article here:
https://www.kansascity.com/news/local/community/cass-county-demo/article221846335.html
NOW Essential Oils – Food Grade? FAQs


Life Thyme Botanicals is excited to be offering NOW Essential Oils!
NOW® Solutions offers high-quality, pure essential oil products that we are proud to offer to our customers of burniva.com. Its important to know the safety facts on essential oil usage. Please use the following link to learn more about NOW essential oil Food Grade?
NOW Food Grade? FAQs
How Comfrey Helped Me

Hi Folks! Emily here. I wanted to share with you an amazing remedy that I have had the opportunity to use today. It’s that time of year where bugs are becoming a big problem. Well yesterday morning I woke up with a massive spider bite on the back of my left thigh. It was swollen and red and hurt like crazy with a deep throbbing pain. I slapped some lavender and peppermint on it to deal with the itching and pain and I went on with my busy day, only giving it thought when it began to hurt again. By the end of the day the bite had swollen and reddened nearly my entire thigh. I applied some lobelia tincture topically (which took away the pain immediately) and took some anti-allergy tincture that I keep on hand and went to sleep. In the past that had always taken care of problems like this. Not so this time. When I woke up this morning I had a mild fever and the redness and swelling had not diminished noticeably. So my trusted standbys had helped the symptoms but not effected a cure. I hustled to the shop and grabbed an herb that I really should have had all along.
Comfrey.

I made a tea out of the leaves and after steeping the leaves for 10 minutes, applied the wet leaves (still warm) to the area of the bite. I kept those on for 5 or 6 minutes and then applied them again every 10 or 15 minutes for an hour (normally I would wrap this poultice in gauze and leave on for several hours but I was in a hurry). See here now. I then discarded the leaves and for the rest of the day I have been dipping a washcloth in the tea and applying it to the spot every half hour or so. It is 3:51 pm as I write this and the swelling and redness has gone down so that my entire leg is back to normal, the fever is gone and there is only a little red spot to mark the original site of the bite or sting or whatever got me. Comfrey has proven its worth to me and from now on I will make sure to always have some on hand!.